Selfcare innovation: in-licensing or custom-made? How to choose the best strategy for your brand?
In a constantly evolving selfcare market, brands must be quick to react and keep up with regulatory changes while staying in tune with consumer trends and expectations. Nurturing and guiding a strong innovation pipeline proves is key to reconciling these challenges and ensuring long term brand performance. There are two main ways to achieve this: in-licensing, and customised development. How do in-licensing and “from scratch” development complement each other to strengthen a brand’s innovation strategy?
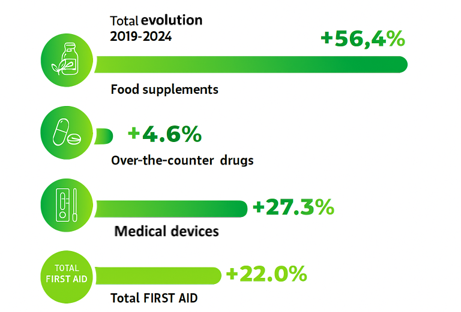
The vitality of the selfcare market is reflected in the sheer number of launches: 1,600 new food supplements hit French pharmacy shelves in 2023, accounting for more than two thirds of first-aid innovations, a category that includes products accessible without prescription (source: Nérès)1.
Selfcare: the race for innovation
Since 2014, innovation in food supplements has increased by +176%, while medical devices still accounted for 68% of new products launched in 2023. This acceleration reflects both the need for brands to remain competitive and the need to meet consumer expectations, while evolving within a demanding regulatory framework. A complex equation which, when it becomes strained, forces players to rethink their approach – as the Boiron case showed after homeopathy was delisted from reimbursements in January 2021.
Adapting and innovating in selfcare in the face of regulatory disruptions
Boiron: a strategic rebound through innovation
The Boiron case illustrates how a historic brand can turn a major constraint into a lever for innovation. The discontinuation of reimbursement of homeopathy in France led to a drastic decline in sales of Homeopathy with Common Name (HNC). In this new regulatory context, the company launched, under the impetus of its new CEO Pascal Houdayer, a strategy giving a central place to innovation. This approach opens new development prospects for Boiron, notably based on the exploration of high-growth categories such as food supplements.
It is within this context Oscillo’ has been launched, a range that aims to be“Simple, practical and designed to support both children and adults throughout the seasons.”2 With Oscillo’, Boiron expanding into the most dynamic food supplement categories, targeting two particularly promising segments: immunity and vitality.

Boiron Oscillo product range – 2025
The laboratory has also initiated a reflection on dosage forms: bilayer tablets, orodispersible sticks, effervescent tablets… practical formats, easy to use and designed to promote adherence (to go further on this topic: Galenic innovation: the value of reconciling efficacy and pleasure in dietary supplements). These products combine naturally sourced active ingredients, vitamins, minerals, and plant extracts, in line with Boiron’s shift toward a broader natural health offering.
Weleda: a deep transformation in the world of selfcare
Another iconic selfcare case is Weleda. A major player in natural and organic health and cosmetics, the group had to contend with the rising number of competitors in natural cosmetics and the delisting of certain homeopathic medicines. However, in 2023, Weleda France marked a recovery in growth, a rebound largely driven by the launch of 27 innovations, notably in facial care and anti-wrinkle products, as well as a notable breakthrough in natural and organic deodorants, where Weleda reached 35% value market share3. The brand thus confirms its ability to spot trends, innovate quickly and adapt its product portfolios.
Weleda is also undergoing a deep transformation. In her interview with Premium Beauty News (2025)4, its CEO Tina Müller explains: “We have transformed our organisation as much as possible to become an agilty driven and digitalised organisation.” She also highlights changing consumer behaviour: “We are seeing a boom in the food supplement market; this trend toward inner and outer beauty is present almost everywhere in the world. […] The premium market is experiencing tremendous growth, driven by the underlying trend of longevity.”
These statements resonate particularly with the new dynamics of selfcare: the emergence of in & out solutions, the growth of the longevity sector and the growing need for hybrid products combining beauty, health and naturalness. Weleda is fully aligned with these movements with the launch of premium ranges such as Blue Gentian & Edelweiss or Cell Longevity, natural cosmetics formulated around a plant-based NAD⁺ booster to act on cellular longevity and the visible signs of skin ageing.

Cell Longevity natural cosmetics product range
Arkopharma: ongoing innovation and product dynamism
A specialist in herbal medicine and natural health, Arkopharma illustrates another model of product dynamism. In an interview given to Actif Mag (No. 91, 2025)5, Aurélie Guyoux, Research and Development Director, indicates that the company launches more than 20 new products per year in pharmacies, continuously strengthening its plant-based health product range.

Arkorelax® Sleep & Anxiety (launched in September 2025). Formulated with a patented lemon verbena extract.
In-licensing & customised development: two strategies to innovate
Innovation is not occasional but structural: it constitutes the very foundation of competitiveness in natural health. It enables brands to move forward and adapt in an increasingly demanding regulatory environment, while responding to rapidly changing consumer expectations: more proven efficacy, scientific soundness and safety guarantees. More than just a growth lever, innovation reflects the brands’ DNA and agility, their ability to remain credible, distinctive and sustainable over time.
In this context, one question springs to mind: how can we innovate effectively while combining speed and differentiation? Between ready-to-market solutions via in-licensing and custom developments, these approaches are now proving to be complementary in building high-performing, value-creating portfolios over the long term.
In-licensing: gaining speed and agility
In-licensing is emerging as a key lever for accelerating innovation and strengthening brand agility. It enables brands to reduce time-to-market, effectively complement in-house R&D capabilities and quickly enrich their portfolio with products already developed and already proven in other territories. Based on a rigorous due diligence process – scientific, technical, regulatory and business – in-licensing makes it possible to integrate high value-added solutions while managing financial and operational risks.
The main advantages of in-licensing:
- Faster access to market, based on already developed solutions complementarity with in-house R&D strengths, without undermining from-scratch innovation capacities
- Complementarity with internal R&D strengths, without cannibalising in-house, from-scratch innovation capabilities
- Rapid portfolio expansion, through products that have demonstrated performance in other geographical areas
- Opening to new territories, through the acquisition of marketing rights
- Reduced financial and technical risks, compared to complete in-house developments
The specific case of medical devices and in-licensing
This relevance of in-licensing is even more pronounced for medical devices, in a regulatory context deeply transformed by the entry into force of Regulation (EU) 2017/745 on medical devices (MDR – Medical Device Regulation), which replaced the former MDD directive.
Under the MDD, launching a medical device was largely based on existing scientific literature and notified bodies validated CE marking through pathways that were less strict than today. It was within this framework that certain industrial players have been able to build extensive portfolios of medical devices, now offered to brands on a licensing or distribution basis.
With the MDR, requirements have changed significantly: more rigourous clinical data, enhanced performance demonstration, increased requirements for risk management and post-market monitoring. In practice, developing a medical device “from scratch” now involves lead times often exceeding two years and significant investments, depending on the device class.
In this context, in-licensing appears as a major strategic lever, enabling brands to access medical devices that are already MDR-compliant, with strong clinical data and secure CE marking, while limiting regulatory, financial and operational risks.
From scratch: customised innovation
Customised development in selfcare is a preferred path for brands wishing to go further in differentiating themselves and in expressing their identity. Unlike in-licensing, where the product and its supporting documentation are by nature already structured, this approach offers total freedom of design, making it possible to shape a solution perfectly aligned with the brand’s DNA. The product is not set in stone: it evolves throughout the development process, through scientific, marketing and sensory considerations, to produce a truly custom-made creation.
The main advantages of from-scratch development:
- Full expression of brand DNA: the ability to bring a distinctive touch, both in terms of substance and in the form of the product.
- Expert and differentiating formulation: a refined physiopathological approach, selection of active ingredients and adjustment of dosages.
- Integration of market insights and consumer experience: joint work on sensoriality, user satisfaction and perceived effectiveness, promoting long-term compliance and adherence.
- Galenic and organoleptic freedom: choice of formats, textures, tastes and smells, enabling the creation of true signature products – such as the iconic scent of immediately recognisable Mustela baby products.
- Control over the level of proof and reassurance: free choice of ingredient suppliers, active ingredients and supply chains, possibility to integrate dedicated clinical or consumer studies, as well as additional analyses guaranteeing quality, traceability and safety of ingredients and raw materials for more information on this topic: Plant-based food supplements: how can a brand secure and enhance its offering?)
Even if development timelines are longer, this approach, particularly suited to food supplements, makes it possible to build unique products, rooted in the brand’s identity and providing long-lasting differentiation.
BOTANIBRANDS, partner of selfcare brands fueling innovation
In an environment where innovation must be both fast, credible and differentiating, BOTANIBRANDS supports brands at every stage of their development, enhancing their portfolios with high value-added innovations.
Whether through in-licensing solutions or through “from scratch” developments, BOTANIBRANDS acts as a true strategic partner, capable of adapting its approach to the challenges, ambitions and DNA of each brand.
BOTANIBRANDS’ proposals from in-licensing and from scratch:
- ready-to-market in-licensing solutions (medical devices, food supplements), selected for their level of innovation, performance and market suitability;
- customised support from A to Z for from-scratch development projects, drawing on regulatory, scientific, industrial and marketing expertise;
- carrying out in-depth due diligences on in-licensing opportunities, to ensure their scientific, technical, regulatory and business content before any decision is made;
- access to a European network of industrial and business partners, offering technical, regulatory and international opportunities.
Already chosen by many selfcare brands in France and in Europe, BOTANIBRANDS positions itself as an innovation catalyst, delivering high-performing, differentiating and sustainable portfolios.
Want to accelerate or structure your selfcare innovation strategy?
Sources :
- Baromètre NèreS des produits de santé et de prévention de 1er recours 2023
- https://www.boiron.fr/nos-produits/oscillo
- Gers, part de marché valeur, déodorants naturels et bio, année 2023, pharma + parapharmacies
- Premium Beauty News « Nous visons un milliard d’euros de chiffre d’affaires », Tina Müller, Weleda – de Kacey Culliney
- Arkopharma, au coeur du végétal pour une effcacité ciblée – Actif’s mag numéro 91